A manufacturer located in Bucks County recently created an important medical implement that was approved by the FDA. John George wrote about the technological advancement for the Philadelphia Business Journal.
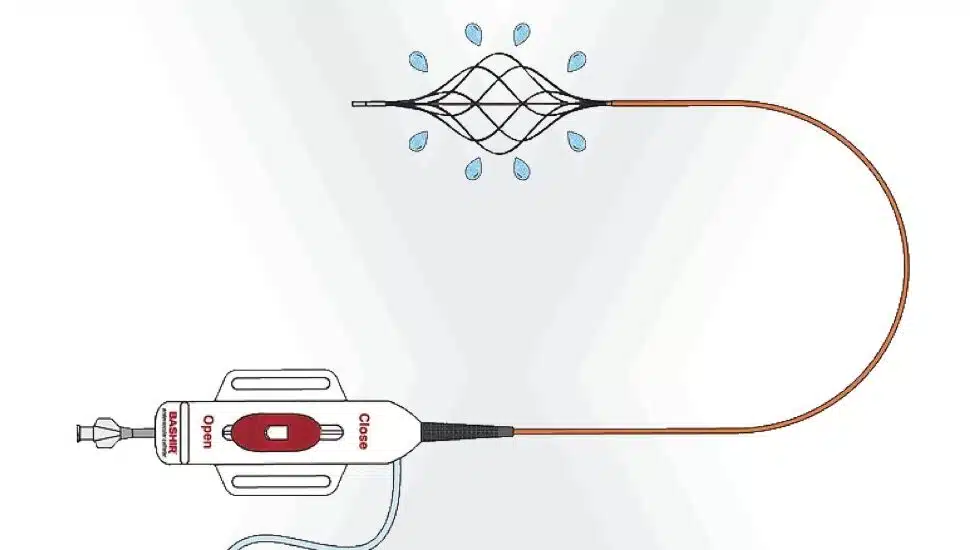
Thrombolex Inc., located at 75 Britain Drive in New Britain, will be marketing the Bashir Endovascular Catheter device, created by the manufacturer’s founder, Dr. Riyaz Bashir. With the assistance of Nicholas Green, Bashir invented the device at the Lewis Katz School of Medicine at Temple University, located in Philadelphia.
The device, which will be used as a treatment for acute pulmonary embolism, was recently approved for use by the Food and Drug Administration.
“This FDA approval is a significant milestone in acute pulmonary embolism treatment,” said Bashir.
Outside of his role with the Bucks County manufacturer, Bashir is a professor of medicine at the university’s medical school and the director of vascular and endovascular medicine at Temple University Hospital.
Read more about the medical tool in the Philadelphia Business Journal.
_____